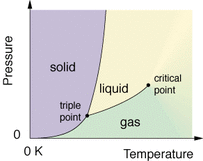
Before beginning the lab, we took some notes while my teacher explained to us what a phase diagram is, where it demonstrates how every substance can either be a solid, a liquid, or a gas depending on the pressure and temperature. Every substance can be at any phase only there's 2 conditions to consider. The first is temperature and the second is pressure(or compression, another form of pressure). My teacher used a pressure gauge, and this wide u-shaped tube to show us how dry ice (frozen CO2), can turn into liquid, and then to a gas. He set the pressure gauge to 80 psi(which is about quarter of the way up the y axis in the graph to the left), and put some dry ice into the tube. Then he closed the tube shut very tightly, so that the top wouldn't pop off with all the pressure and hit someone. After tilting the tube left and right a couple times, the CO2 started to turn slushy, and then it turned into liquid. Once it turned into liquid, he unscrewed the top off and the used-to-be liquid exploded in a mess of gaseous CO2. After demonstrating that, he began to tell us the directions for the mini-lab we were about to have.
The first step to this experiment was to drop a small piece of dry ice inside a beaker of H2O, then comment/observe the difference in amount of solid compared to gas. The second step was to place a small piece of frozen CO2 (1 cm in diameter) into a balloon and tie it off. Then, measure the diameter in the balloon once it completely sublimes.
The purpose for this experiment was to see for ourselves how matter can change from solid, to liquid, then to gas, and have a general understanding of what a solid and a gas is by using solid CO2 and H2O. I worked with Morgan, Jennifer, and Nancy for this lab. We grabbed a beaker and filled it with water from the tap. Then we dropped some dry ice into the beaker. Immediately, the gas from the dry ice started rising from the beaker. Inside the beaker, you could see a bunch of bubbles popping up from the dry ice piece(as seen below).
The first step to this experiment was to drop a small piece of dry ice inside a beaker of H2O, then comment/observe the difference in amount of solid compared to gas. The second step was to place a small piece of frozen CO2 (1 cm in diameter) into a balloon and tie it off. Then, measure the diameter in the balloon once it completely sublimes.
The purpose for this experiment was to see for ourselves how matter can change from solid, to liquid, then to gas, and have a general understanding of what a solid and a gas is by using solid CO2 and H2O. I worked with Morgan, Jennifer, and Nancy for this lab. We grabbed a beaker and filled it with water from the tap. Then we dropped some dry ice into the beaker. Immediately, the gas from the dry ice started rising from the beaker. Inside the beaker, you could see a bunch of bubbles popping up from the dry ice piece(as seen below).
We left the dry ice inside the beaker while we did step 2, dropping a piece of dry ice into a balloon, and then tying the balloon off. We grabbed one of the bigger balloons and dropped a piece of dry ice that was 5 cm in diameter instead of 1 cm. This time, we left the balloon while we went back to experimenting with the CO2 and H2O. I occasionally glanced at the balloon to see it slowly growing larger and larger, with the gas coming off the dry ice, inflating it(as seen below).
When I was watching the dry ice and the gas rising from the beaker. I most intrigued by the gas, it's just cool to look at. I grabbed a funnel that was sitting on the table and placed it on top of the beaker, the tube side facing up. The only place for the gas to go was up into the tub-y part of the funnel. The gas came up in little circular puffs of smoke. It reminded me of Absulum the Caterpillar from Alice in Wonderland, with his little circle shaped puffs of smoke. Unfortunately, I don't have a picture to post of it, but I did see Nancy taking pictures of it so I'll ask her.
Andrew came by and he showed us how to make bubbles and something else. He used this thin little telescope looking tool to make the bubbles. He showed us that if you dip it into a mixture of glycerin and normal dish soap, and you make the bubble. Then you carry the beaker that has the dry ice and H2O in it to the bubble that's still floating in the air, (and you successfully avoid popping the bubble)the bubble will stick to the top of the beaker and the gas rising from the dry ice will, instead of just rising into the air, will go into the bubble(as seen below). Then the bubble just sits there until you pop it and the gas gets let out(as seen below).
I was surprised that getting the bubble to sit on top of the beaker was probably the hardest part because, well, it's just blowing a bubble. It should be easy to do but this time it wasn't. Morgan wanted to see what would happen if you put the soap into the beaker with the dry ice. Once you did that, a whole bunch of bubbles kept on rising and dripping down the side of the beaker(as seen below). I didn't expect so many bubbles to just rapidly rise out of the beaker. I thought the top of the water was just going to be bubbling a lot when Andrew mentioned to avoid putting soap in the beakers.
After experimenting with the dry ice and bubbles, we measured the diameter of the balloon once we knew that all of the CO2 was in its gaseous phase of matter. It ended up being 22 centimeters in diameter.
The reason for that specific instruction became clear after everything was put away and cleaned up. Andrew showed us an equation that could give you the ratio of a solid piece of CO2 to gas of the dry ice.
4/3 x π x r^3
Solid CO2 Volume
4/3 x 3.14 x (2.5, 2.5, 2.5) = 65.416
Balloon Volume
4/3 x 3.14 x (11, 11, 11) = 5, 572
Ratio from solid CO2 : gas of the dry ice
65.416 : 5,572
I found this lab very entertaining and I'm very glad I got to do it because now I have a better understanding of the phases of matter then I ever have before. From doing this lab, I learned what a phase diagram is and how it works. What a pressure gauge is because I've never seen it before and now I know what it can do. I learned a neat way of inflating balloons without having to waste a breath. I've never put dry ice in water before so now I can say I have done that. I learned an awesome way to make bubbles. And finally, I learned the workings of the three phases of matter.
Here's a video I took, with Jennifer's phone, of some of the lab we did:




 RSS Feed
RSS Feed
