|
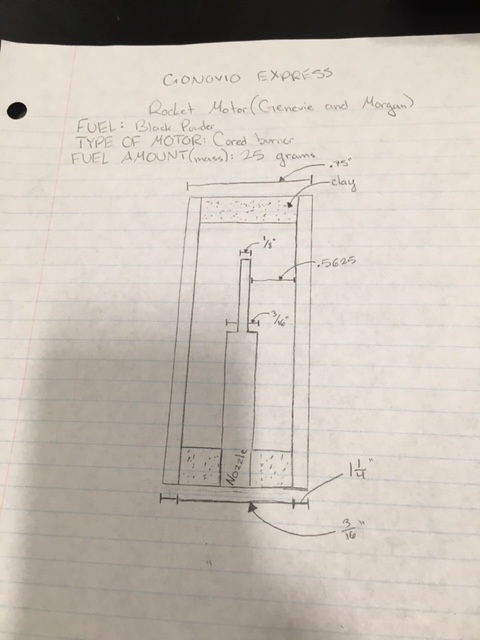
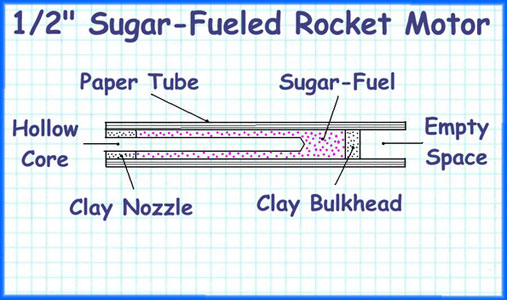
This is our blueprint for the motor. Morgan and I chose to do a cored burner with balck powder fuel.
0 Comments
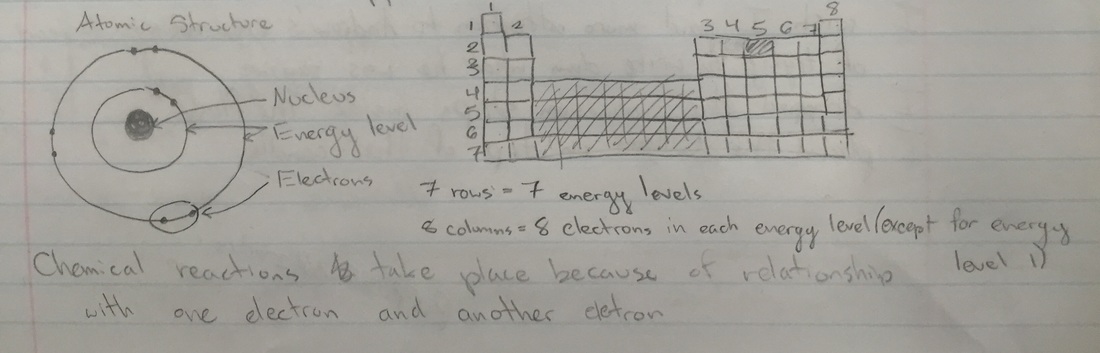
Background Research CHEMICAL REACTION: Taking some substances and making them into new substances. Or as Andrew says, "Taking some stuff(atoms) and turning it into new stuff." A change in matter where the atoms in one or more substances combine, separate, or rearrange to form one or more new substances. HOW DO REACTIONS HAPPEN? Atomic structure which is the nucleus, energy level and electrons. Rows tell you the energy level, and the columns show the number of electrons on the outer shell. Once the atoms combine and get enough electrons, they are stable. If they are not stable, what happens is the atom is combining with anything possible. ATOMS: You can look at the periodic table and see how many electrons are on its outer shells. COMBUSTION REACTIONS: Substances combine with oxygen to produce water and CO2. RULE OF THUMB FOR CHEMISTRY: Elements combine in such a way, to get a full outer shell of electrons. THERMODYNAMICS OF CHEMICAL REACTIONS: Reactions absorb or emit energy/heat. The substances undergo reactions to reach longer energy states. Energy change is predictable. "I want to be the catalyst for this." ---> "I want to speed this up." Performing the Lab Top-Left: That was after the citric acid was poured into the volumetric flask. Top-Middle: These are the magnesium strips. Top-Right: This picture was taken after we dropped the magnesium strips inside the flask and capped the flask with the balloon. We had to curl the strips so they could quickly and easily drop into the flask. The citric acid dissolves the magnesium strips because citric acid dissolves metal. And we capped the flask quickly with the balloon so as not to let the dissolving magnesium strips release any hydrogen that won't go into the balloon. Middle-Left: This is the magnesium strips dissolving because of the citric acid. Middle-Middle: The balloon began to blow up with the hydrogen coming from the magnesium strips. Middle-Right: The balloon inflated more and more. Bottom-Left: The magnesium strips were bubbling a lot and the glass of the flask above the citric acid level was really hot. Bottom-Middle: This is when the bubbling from the magnesium strips was close to ceasing to a stop. Bottom-Right: This is when the bubbling from the magnesium strips, that were dissolved now, completely stopped. This is what the balloon looked like after it completely stopped inflating from the hydrogen and it the end was tied off. We had to hold the ballon down so it wouldn't float up to the ceiling. I think the lab ended successfully, after the magnesium strips were dropped into the flask, the water started to fizz up and the balloon began to inflate with hydrogen just how it was supposed to happen. Once the balloon was tied off and the excess of the tie was cut off. We taped it onto the end of a long stick. While I lowered the balloon onto the candle that was sitting on the table. Once the balloon reacted with the flame, we felt the balloon and it was wet, which was the water created when the balloon combusted. Morgan took a slo-mo video of the combustion. Skip to 0:30 if you only want to see the balloon combusting (looks really cool!). Most of my questions concern the background research we did. I was confused about how you're supposed to find how many electrons are in an element using the periodic table of elements.
I wanted to do this lab because Andrew said it would give us a little bit of a deeper understanding of what's happening inside a rocket motor, in preparation for making a motor. I think I performed well in this activity. I had simple responsibilities like dropping the magnesium strips inside the flask and lowering the balloon at the end of the stick onto the candle flame. Since, they were simple, they were easy to follow through on. Although I do wish I paid more close attention to Andrew's explanation of the electrons in an atomic structure instead of trying to write down what he was saying. Had I done that, I think I would've understood it better. I worked with Morgan in this activity, because we've worked well together in past projects. I think as partners, we performed well because we shared the responsibilities of the lab and made sure to follow the instructions given to us. I don't think I would have changed anything in this activity to get better results. The activity went smoothly without any complications. I think I'd like to do another experiment which includes hydrogen. Fire just adds more enjoyment in labs, it's exciting. Although I don't really know what labs we could do that have fire but haven't already been done. Guided activities work pretty well because learning the background research is a lot quicker and there's no risk of not getting it done in one day because of things such as insufficient resources, or time management. Black powder is made from Potassium Nitrate (KNO3), Charcoal and Sulfur. Black powder can be used as a propellant with more consistency than zinc-sulfur and is the propellant used in small commercial model rocket engines. The burn rate of black powder can be slowed by the addition of other components. The sugar-rocket fuel consists of three simple ingredients:
To facilitate ignition, a small dimple is formed in the grain’s rear end. just big enough to accept an igniter. When properly made, end burners produce a constant level of thrust for a comparatively long time. But they have the disadvantage that the inside wall of the casing is exposed to open flame for the duration of the burn. Because the casings for the motors in this topic are made of paper, the implications are obvious. If you make the burn time too long, you’ll burn through the casing wall. Also, for the chamber pressure and thrust to remain constant, it is absolutely imperative that an end burner’s propellant burn at a perfectly uniform rate. The flame in an end burner travels in a straight line from the point of ignition to the forward bulkhead. The dome-shaped flame front, formed immediately after ignition, generates a surge of high pressure and high thrust. As the dome changes into a shallow cup. both chamber pressure and thrust are reduced, and thereafter remain constant. A cored rocket motor is the traditional motor, typically with a clay nozzle, and a hollow core going up through the nozzle and into the fuel grain for some distance. Ignition starts at the core’s forward end. and instantly spreads to the rear, lighting the length of the core as it goes. From that point on the flame spreads outward in the shape of an expanding cylinder, consuming the last of the propellant as it reaches the inside of the casing wall. In motors with small nozzles and short cores, the chamber pressure and thrust start out low and reach their maximums at burnout. In motors with larger nozzles and longer cores the initial values are higher. In motors with very large nozzles and very long cores the starting pressure and starting thrust can exceed 50% of their maximums.
A motor’s core length is critical because it directly controls the amount of propellant burning at any given moment. If the core is too short, the motor performs poorly. If the core is too long, the maximum chamber pressure can exceed the motor’s design limits, and cause the motor to explode. Because the burning surface in a core burner is larger than the burning surface in an end burner, a core burner requires a slower burning propellant. Because the distance that the flame travels is short (from the edge of the core to the inside of the casing wall), small core burners have comparatively short burn times. All core burners have the advantage that the layer of unburned propellant between the flame and the casing wall insulates the casing from the heat of the flame until the final moments of the burn. Week 1
10/19, Monday: Plan the next 2 weeks 10/20, Tuesday: Research candle making 10/21, Wednesday: Candles 10/22, Thursday: Rock Research(Phase 1) 10/23, Friday: Rock Research(Phase 1) Week 2 10/26, Monday: Staff Day(No School) 10/27, Tuesday: Guided Activity(50 pts.) 10/28, Wednesday: Rocks(Phase 2) 10/29, Thursday: Rocks(Phase 2) 10/30, Friday: Blog Posting The link to the science article I read is located above.
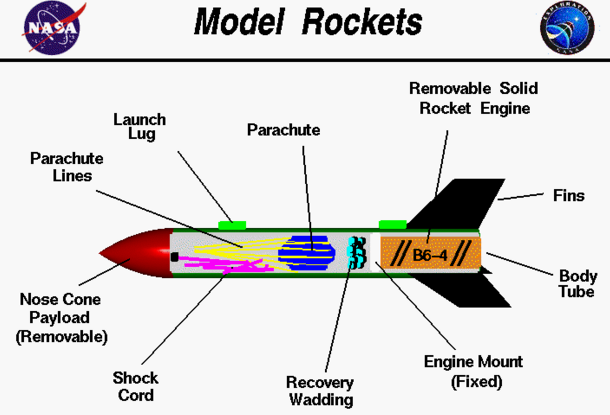
The topic for this article is wormholes. Scientists today are searching for traversable wormholes. To keep a wormhole open long enough to traverse it would require some type of scaffolding, but regular matter would not be enough, you would need some type of "exotic material." Dark energy and dark matter is said to be considered "exotic material." The negative pressure of dark energy produces the gravitationally repulsive force that pushes the space inside our universe outward, thus producing the inflationary expansion of the universe. Dark matter is five times more prevalent in the universe than regular matter. Although scientists cannot detect dark matter or dark energy, they can still still learn about these materials by examining what effects it has on the space around them. Like in Interstellar, nothing would happen to someone passing through a wormhole, there would be no "intolerable gravitational tidal forces acting upon the spacecraft or its passengers." Travelers would appear as if they're traveling faster than light. Something that surprised and that I found interesting was when it was said that based on certain wormhole theories, Ali Övgün of Eastern Mediterranean University in Cyprus compared peering through a wormhole to Alice's glimpse through the looking glass, in Lewis Carroll's novel of the same name. I think it would be really awesome to see a wormhole that reminded you of Alice's glimpse through the looking glass. I'd feel like I was actually in Alice in Wonderland! When I picture a wormhole I think of what the wormhole looked like in Interstellar This was the rocket project that we did as a class. I chose to participate in it because I know that I have done rocketry in the past, but this was a new kind using new materials, so I was interested in learning about it and trying it out. I think that our rocket was one of the most successful ones that the class launched. To launch our rockets we took a trip to fiesta island during chemistry class because that is one of the few places we are legally allowed to do it. When we were launched ours there was a small problem at first. The launch lug, which keeps the rocket straight when its launched, was getting caught and causing too much friction. So Andrew, spent a little bit of time getting them to work correctly. But we did the actual launch ours worked perfectly and actually went very high and drifted far, as the parachute came out and worked as planned. The very first thing you had to do for model rockets is choose a partner. I chose to work with Morgan for model rockets because we've worked well together in past experiments. The second we had to do was familiarize ourselves with rocketry and the components listed in the image below: After Morgan and I researched rocketry and the components listed above, we discussed what we found to make sure we were on the same page. Then we went to ask Andrew for the model rocket, but before that, he asked us some questions about rocketry so he knew we were ready. After that, we started making the model rocket using the instructions the kit provided. During the construction of the model rocket, we came across a few minor problems. The first came from placing the engine inside the bottom of the rocket. I guess the engine mount was too close to the bottom because the engine wouldn't fit in all the way, with the end of the engine sticking out of the rocket a little bit. The second problem we had, which wasn't that bad but still kind of worried me in the beginning was how the parachute and the shock cord had a hard time staying inside the rocket. We had to press the nose cone into the rocket to make sure it wouldn't fall off and the parachute and shock cord would fall out too. I was also afraid that the nose cone would get pushed by the wind in the sky after it was launched and the parachute would pop out too early. I guess this was an irrational fear though because if the nose cone is piercing through the sky, then surely the wind slamming into the nose cone would keep it from disconnecting from the rocket. The third and final problem we had was when the rocket was being readied for launch. The launch lug, which keeps the rocket going straight when it's launched, was getting caught and it caused too much friction. So Andrew spent a little bit of time getting them to work correctly. Eventually he was successful and once the rocket was launched it went really high and drifted far. The rocket did better than expected so I'm pretty happy about that. This is the model rocket we built, it has all of the necessary components; a parachute(which was tucked into the rocket after the recovery wadding was placed inside and capped of by the nose cone), a launch lug, the rocket engine, fins, body tube, the fixed engine mount, the recovery wadding, and the shock cord.
I'm glad I had this experience because I gained more knowledge on rockets than I had before. Even from making rockets in 8th grade, I can only remember so much, like the thrust of the rocket, what do the fins and nose cone do for the rocket, etc. I learned more about the actual components of a model rocket. I didn't know that you needed a launch lug before making the model rocket. I thought that the purpose for the shock cord and the recovery wadding was pretty interesting and clever. It's cool that there's parts of the rocket like those components that take care of problems that might occur. Like the recovery wadding prevents the parachute from burning up because of the engine going off. Doing the Lab Here are all of the supplies we used for the experiment; citric acid, baking soda, cornstarch, olive oil, essential oils, water, a bowl, gloves, and a cupcake mold. We started by pouring the necessary amounts of citric acid, baking soda, cornstarch, and olive oil into the bowl. We didn't have any measuring cups so we had to use a tablespoon, we converted cups to tablespoons to ensure that we were adding the right amounts. For the olive oil, where the necessary amount was teaspoon, we just estimated that the tablespoon we used was 1/3 full with olive oil. Throughout pouring, we stirred the mixture with our gloved hands to make sure all of the ingredients were mixed thoroughly. After we added the citric acid, baking soda, cornstarch, and olive oil to the bowl, we gradually poured water into the bowl, just enough so the mixture was compact. Once we added water to the mixture, with only one of our gloved hands (by this point, one of each of our pairs of gloves were dirty from touching the ground) we tossed the little ball of mushy mixture before pressing it into one of the cupcake molds. After having 5 cupcake molds filled by the mixture we cleaned up and left it to dry in the chemistry class. Placing the Bath Bombs in Water The bath bombs looked kinda weird. They looked bubbly and solid. I don't think that's what they were suppose to look like. Although the bath bombs did fizz a little when we placed it in the water so it did work a little bit. When the bath bombs were placed in the water they didn't really stay solid and keep fizzing. They fizzed for a couple seconds and then they dissolved in the water so they didn't stay solid at all. I kind of expected something weird like this to happen since the bath bombs looked bubbly-solid inside the cupcake mold and I was sure they weren't suppose to look like that. What happened is that when the citric acid and sodium bicarbonate are exposed to water they react by fizzing which produces carbon dioxide mixing the other ingredients in the bath bomb with the water. This sort of happened, it started out fizzing but the fact that the bath bomb started to dissolve and fall apart ruined the reaction.
Morgan and I think that the reaction didn't go as planned because we added the water too fast to the ingredients. By adding water to the ingredients too fast, instead of adding it little by little, we exposed the ingredients (baking soda, cornstarch, citric acid, and olive oil) to the water too much, strongly reacting to the water. If I redid this experiment, I would pour the water into the bowl slowly instead of pouring it fast like we did today. I would also make sure we have food coloring because this time we couldn't find any so our bath bombs weren't colored. I think I performed well in this activity. I didn't do anything to prevent the experiment from happening, like forgetting to bring an ingredient. Although I do wish that Morgan didn't have to get a majority of the ingredients, I just didn't really have most of the ingredients at my house. I told my mom that we were making bath bombs in chemistry and she said that I should save one of the bombs we made for her. But since the bath bombs didn't really work out very well, I think that since I know how to make them now, I can make bath bombs for my mom in the future. The purpose of this lab was to learn more about measuring stuff. The lab focused on these questions: How can we measure something lighter than air? Can we measure the density of the helium inside the balloon? Background Research Background in Scientific Measurements Length: meter (m) 4m = 4 meters megabyte - gigabyte - terabyte - pitabyte Volume: Time: seconds (s) Amount of stuff: Mole (mol) "stupid large" like the number of atoms Temperature: Kelvin (K) conversion: degrees C + 273.15 = k Secondary Measurements: Ratio + Comparisons Velocity: distance/time = d/t = m/s If you know one of them then you know the other Molarity: mol/L Density: mass/volume = g/m a measurement of how much stuff is in a given volume buoyancy: the level to which 2 substance's densities match/relate negative buoyancy, positive buoyancy, neutral bouyancy What We Did Andrew showed us that a balloon has positive buoyancy(helium) and air has negative buoyancy(gravity). The balloon has volume and the air has mass. Air has a density of .001225 g/cm^3. To begin the lab, we filled a small balloon with helium then measured the diameter of the balloon. We wanted to make the balloon as buoyant with the air as we could so we added some negative buoyancy, or mass, to the balloon, which was a piece of tape sticking to the balloon. Diameter = 17 cm Radius = 8.5 cm We plugged the radius into this equation: Volume of a sphere = 4/3π(8.5)^3 = 2572.44 cm^3 Then we took the density of air and the answer to the volume of a sphere to make this: .001225g --------- (2572.44 cm^3) 1cm^3 We crossed out cm^3 and divided .001225g by 2572.44 = 3.151239g = 3.151g So at this point we knew that the balloon weighed 3.151g. But 3.51g is the weight of not only the helium inside the balloon, but also the balloon itself and the tape that was stuck to it. To find the weight of only the helium, we had to subtract the weight of the balloon and the tape from the overall answer we got before(3.151g). We put the balloon (after it was popped) and the tape onto a gram scale but the scale wasn't sensitive enough so we had to estimate(2.5g). 3.151g - 2.5g = .65g At this point we finally had the weight of just the helium, .65g. The experiment turned out pretty well, I was able to better understand positive, negative, and neutral buoyancy more clearly. Although I think the experiment could have gone better if we had sensitive enough scale, it would have made the math we did more accurate. A little bit after we realized we couldn't find the weight of the balloon and the tape, I asked the question, "What if we just added a heavy object to the scale along with the balloon and the tape, then afterwards, we subtract the weight of the object from the overall weight of the object, the balloon, and the tape? So in the end we would have the weight of the balloon and the tape." Although I didn't think about how the scale wouldn't be able to measure the difference between one gram and half a gram. If I could redo this lab, I would use a bigger balloon because if we had a bigger balloon, then more tape would have been needed. Therefore, we would have more weight from the balloon and the tape than we had in this lab. I think I did well in paying attention to Andrew telling us the background research and I was able to blog more about the lab. I learned that it's important to think of solutions to problems you could have in the experiment. Like knowing the 3.151g wouldn't be the weight of the helium inside the balloon, and knowing that you can fix that problem by just subtracting the weight of the balloon and the tape from the overall weight you got before. I'm going to definetely use what I learned to get around problems in future experiments. |
AuthorHey there! I'm currently in 10th grade at High Tech High in Point Loma, San Diego, California. This is my first year in Chemistry 1, and 2 and this is my class blog. You can learn more about who I am in my 'About Me' page. Archives
November 2015
Categories |


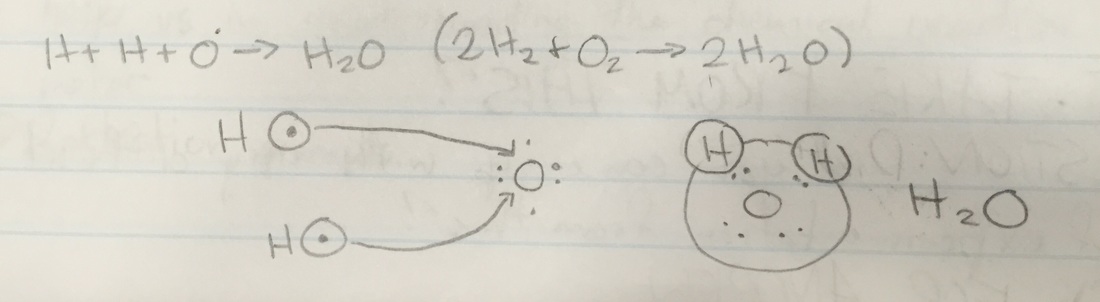
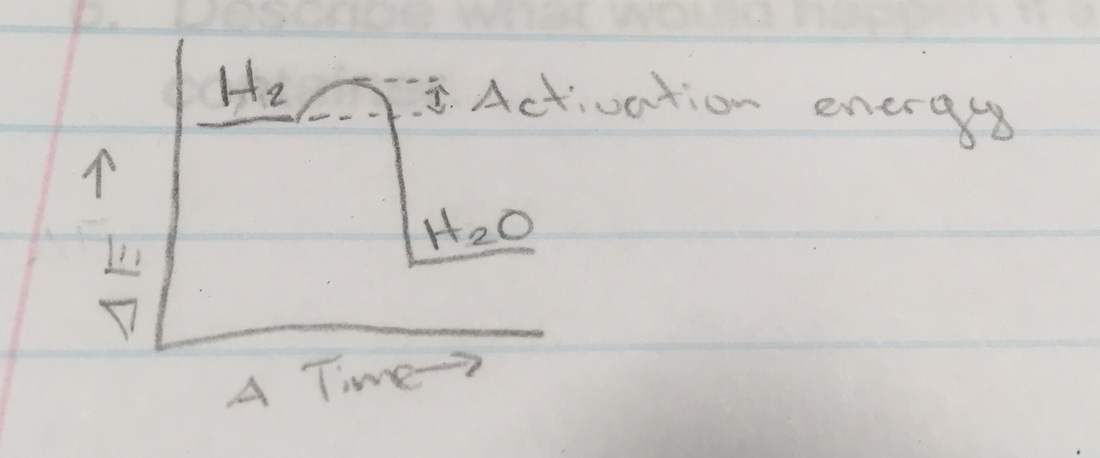
























 RSS Feed
RSS Feed
