To prepare for this lab, Morgan and I researched ionic compounds, like any kind of salt. There is a large variety of salts to use, but after researching how these types of salts will transform into different-looking crystals, we wanted to use a type of salt that will turn into crystals that we would both like to see. In the end, we decided to use Epsom salt.
The supplies I needed for the crystallization lab (which I did with Morgan), were Epsom Salt, blue dye (we wanted to see if the crystals would actually turn blue), water from the tap, a spoon to pour the Epsom Salt inside, and a stir-rod to help dissolve the salt. We used an Erlenmeyer flask to dissolve the salt in the water and then after the salt couldn't dissolve into the water anymore, we poured what was in the flask into a normal cylinder shaped beaker.
We began by filling the Erlenmeyer flask 1/3 of the way with water from the tap. Then we dropped some blue dye into the flask. We placed the flask onto the hot plate (the heat temperature was set at 5) then we poured some Epsom Salt into the flask using a spoon Andrew let us borrow.
We stirred the Epsom Salt to help it dissolve. By turning up the heat temperature, we waited for the thermometer to go up to 70-80 degrees when we placed it inside the flask.
Once it got to around 75 degrees Celsius, we took it off the hot plate and wrapped some parafilm on top of the beaker.
We came back the next day and saw that the dissolved epsom salt didn't turn into crystals at all. In fact, it looked exactly the same as when we placed it in the fume hood on the day before. Andrew said that we could dump out some of the water and pour some more Epsom salt inside the beaker to ensure that we will get crystals when we return the next day.
Sure enough, we did.
We came back the next day and saw that the dissolved epsom salt didn't turn into crystals at all. In fact, it looked exactly the same as when we placed it in the fume hood on the day before. Andrew said that we could dump out some of the water and pour some more Epsom salt inside the beaker to ensure that we will get crystals when we return the next day.
Sure enough, we did.
On Friday, we decided to leave the water with the dissolved Epsom salt water along with the already-formed crystals inside the beaker over the weekend. When we came back Monday, this is what the beaker looked like with all of the left over salt water gone.
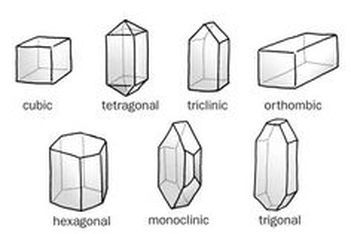
I think the outcome turned out really well, it looks like the Epsom salt crystals I found on Google. AND the crystals are actually blue, although it might just be because there's blue dye in the water that stuck at the bottom of the beaker under all of the crystals (considering how the crystals are a little transparent). The crystals, as seen in the image above, look spiky and jagged, scattered everywhere in the beaker on top and below each other. On a molecular level, the atoms and molecules are arranged in a consistent, repeating pattern, creating one of seven geometrical shapes (as seen below).
I did further research and this is what I found: The small crystals that formed in the saucer grew because of nucleation. A few alum molecules found each other in the solution and joined together in a crystal pattern. Other alum molecules continued to join them until enough molecules gathered to become a visible crystalline solid. (Chemists call that a crystal "falling out of" the solution.)
I think that Morgan and I would agree when I say that we did get the desirable results. Maybe not the first time we tried the lab (when we came back the next day and there was, still, only water in the beaker), but definetely the second time. I think if we had declared the lab finished on Friday instead of Monday then we would have had less crystals in the beaker. Instead of doing that, I suggested that we leave the beaker in the fume hood for the weekend and Morgan agreed.
I think I worked pretty well in this lab. When I worked with Morgan I made sure that we were sharing the responsibility of doing the lab, meaning that I think neither one of us did less than the other. I did an experiment like this one in 3rd grade, I made a snowflake-shaped crystal like the ones Mayra and Isabel made. Back then, I only cared about making a pretty ornament for my christmas tree but now I actually cared about the learning experience.
I wish that we added more sugar to the water the first time we performed the lab so then we wouldn't have had to wait more time for the crystals to form. Although I am still glad that it did happen because then I wouldn't have known that the crystals don't form if there isn't enough dissolved salt in the water. I learned how to make crystals using only salt and water (not considering the other materials needed), which I previously didn't remember how to do it when I made my crystal ornament in 3rd grade. It felt very accomplishing at the end of the lab when I saw all the crystals squished together inside the beaker!












 RSS Feed
RSS Feed
